APPLICATIONS:
- Anti-tumor therapy
- Shows promise for increasing the radiation sensitivity of breast cancer cells
ADVANTAGES:
- In vivo studies have demonstrated the effectiveness of ß1 integrin antibody for anti-tumor therapy
- In vitro studies show enhancement of radiation sensitivity
- The tissue environment responds to cancer in a consistent manner, providing a reliable mechanism for treatment (compared to the variable routes taken by cells to become cancerous)
- ß1 integrin inhibitory antibody had little negative effect on a non-malignant cell line
- 3-D cell cultures can be used to distinguish tumors that are susceptible to the Berkeley Lab treatment
ABSTRACT:
Most current research on cancer initiation and progression focuses on the genetics and the clonal expansion and acquisition of abnormal signaling pathways that characterize the tumor. What has been relatively overlooked is the microenvironment – specifically cell-extracellular matrix (ECM) interactions – and the role that these interactions play in tumor progression and development of resistance to treatment.
Integrins are an important class of molecules that mediate crucial signals between cells and the ECM. There is a growing body of evidence, including clinical trials against specific integrins, showing that interfering with these signals by inhibiting integrin activity can profoundly affect tumor development. Research has shown that ß1 integrin specifically modifies response to chemotherapy in lung, leukemia, and colon cancers. It has also been shown to play a critical role in expression of the breast cancer phenotype (Weaver et al., 1997).
Berkeley Lab Scientist Mina Bissell and University of California at San Francisco Radiation Oncologist Catherine Park, M.D. have now demonstrated that disrupting abnormal cell-ECM interactions by inhibiting ß1 integrin is an effective mechanism for advancing breast cancer therapy and shows promise as a means of enhancing the therapeutic efficacy of ionizing radiation (IR).
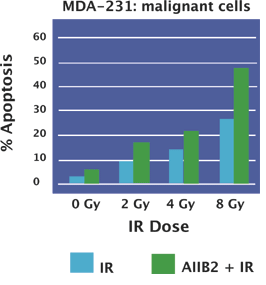
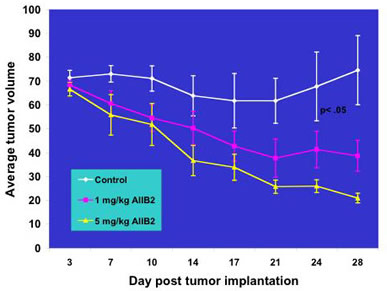
Studies with mice implanted with invasive human breast cancer cell lines show that ß1 integrin inhibitory monoclonal antibody (AIIB2) inhibits formation of tumors and significantly enhances apoptosis and decreases proliferation, and it does so in a dose-dependent manner with minimal toxicity to animals. In addition, when 3-D cultured malignant cell lines are treated with IR combined with AIIB2, the effects are enhanced by up to 50% compared to the use of IR alone. Treatment with AIIB2 resulted in only minimal apoptosis in non-malignant cell lines that have undergone differentiation in a 3 –D model, indicating that treatments focused on inhibiting ß1 activity might have little negative effect on healthy cells.
REFERENCE:
STATUS:
- Published Patent Application # 12/575,411 available at www.uspto.gov. Available for licensing or collaborative research
FOR MORE INFORMATION:
REFERENCE NUMBER: IB-1965
SEE THESE OTHER BERKELEY LAB TECHNOLOGIES IN THIS FIELD:
