APPLICATIONS OF TECHNOLOGY:
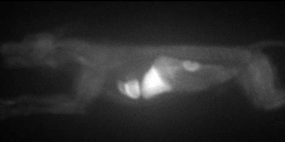
Planar image of iodorotenone in a dog heart.
- PET and SPECT tracers, especially for myocardial perfusion imaging
ADVANTAGES:
- Extraction, retention, and uptake superior to 201Tl, 99mTc-sestamibi, and 82Rb, resulting in higher image quality
- A variety of half-lives including long-lived tracers suitable for delivery to remote imaging sites
- Animal studies show no related toxicity
ABSTRACT:
Researchers at Berkeley Lab have developed rotenone analogs with tracer kinetics superior to 201Thallium,99mTechnetium sestamibi, and 82Rubidium. The rotenone compounds significantly improve the correlation between myocardial tracer deposition and blood flow, resulting in more accurate assessment of cardiac profusion and diagnosis of ischemic heart disease. The Berkeley Lab tracers include rotenone analogs made with isotopes of fluorine, bromine, and iodine having a variety of half-lives. Some are ideal for SPECT imaging, some have half-lives long enough for delivery to remote PET imaging sites, and some have the potential for on-site generation.
All deposited flow tracers currently in use are incompletely extracted and retained by the myocardium, affecting the degree to which the resulting images accurately depict ischemia. Henry VanBrocklin and James O’Neil have demonstrated that the extraction, retention, and uptake of iodorotenone and fluorodihydrorotenone in the isolated, retrograde blood-perfused rabbit heart model are superior to 201Tl, and 99mTc-sestamibi,. In addition, distribution studies of iodorotenone and other labeled rotenone analogs in rats, dogs, and sheep have demonstrated high heart uptake and low lung accumulation with rapid clearance from the blood pool. Toxicity testing of iodorotenone in rabbits given at 100 times the expected human mass dose revealed no effects.
Rotenone is a specific, high-affinity inhibitor of Complex I, a primary enzyme in the mitochondrial electron transport chain. Since rotenone inhibition defines the activity of Complex I, defects in radiotracer binding can be expected to reflect functional changes in the enzyme, and therefore, abnormalities of the mitochondrial energy metabolism. Since the myocardium is rich in mitochondria, rotenone analogs are ideal for clinical evaluation of myocardial blood flow.
Anatonic Physiological Fusion 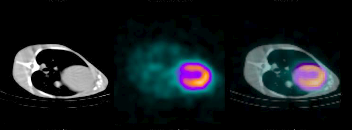
Iodine-123 rotenone SPECT/CT images of canine heart.
STATUS:
- Published Patent Application WO/2008/133730 available at http://www.wipo.int and US-2009-0136424-A1 available at USPTO. Available for licensing or collaborative research.
FOR MORE INFORMATION:
REFERENCE NUMBER: IB-2084, 2085
SEE THESE OTHER BERKELEY LAB TECHNOLOGIES IN THIS FIELD:
